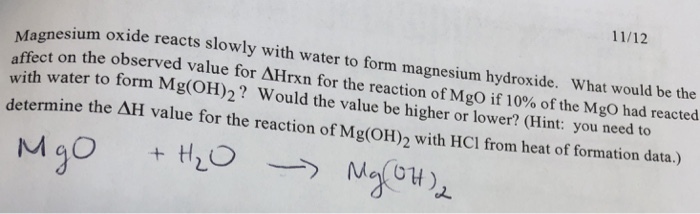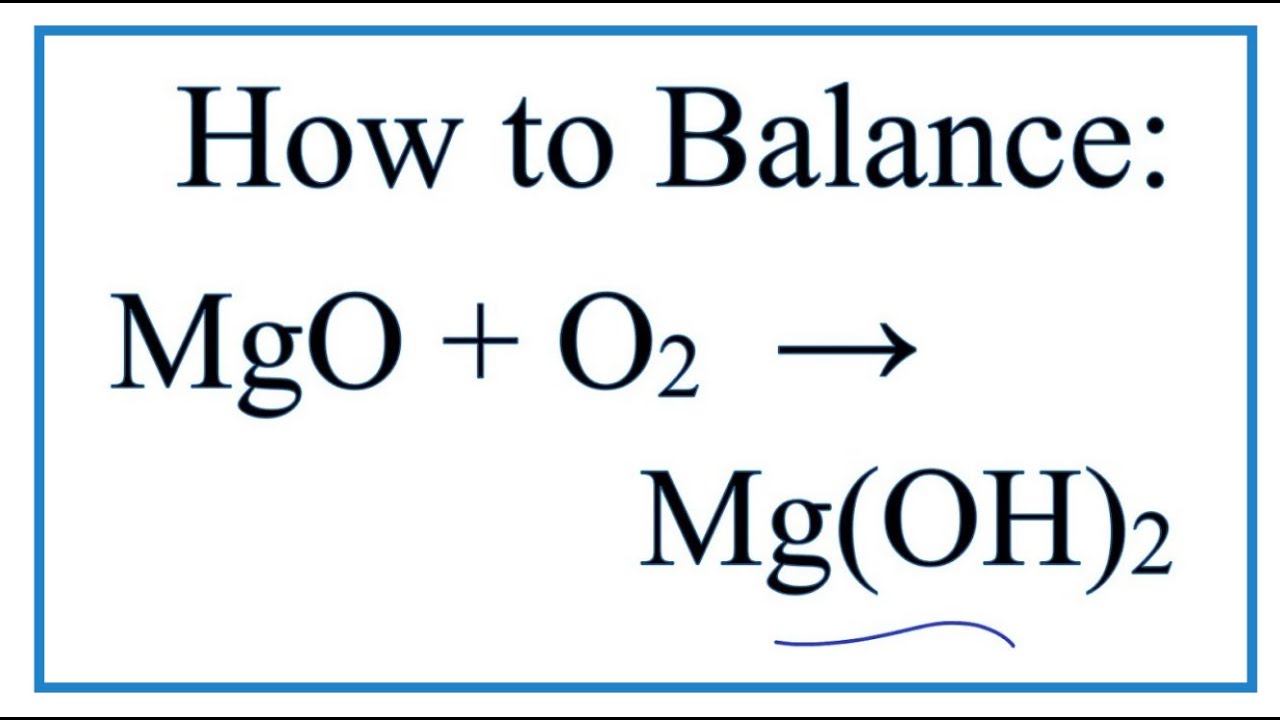SOLVED:Discuss the interpretation of the observation that magnesium oxide is more soluble in aqueous magnesium chloride than in pure water,

When MgO is dissolved in water Mg(OH)2 is obtained the solution thus obtained is ______ in nature - Chemistry - Materials Metals and Non-metals - 16241463 | Meritnation.com
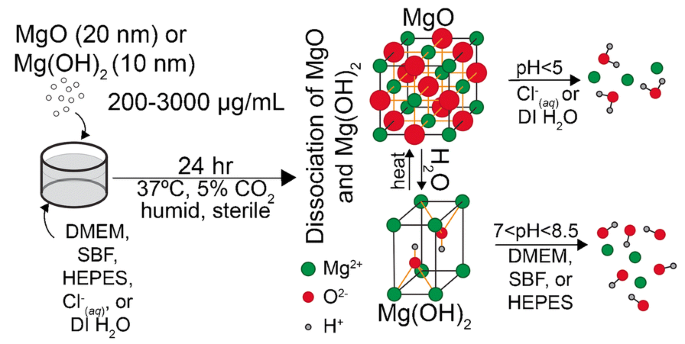
Dissociation of magnesium oxide and magnesium hydroxide nanoparticles in physiologically relevant fluids | SpringerLink

SOLVED:Magnesium carbonate, magnesium oxide, and magnesium hydroxide are all white solids that react with acidic solutions. (a) Write a balanced molecular equation and a net ionic equation for the reaction that occurs