18.4 g of a mixture of calcium carbonate and magnesium carbonate, on heating, gives 4.0 g of magnesium oxide. The volume of CO2 produced at STP in this process is:

SOLVED: The active ingredients in a tablet of a popular antacid are listed as 585 mg of CaCO3 and 112 mg of Mg(OH): These each react with hydrochloric acid found in your

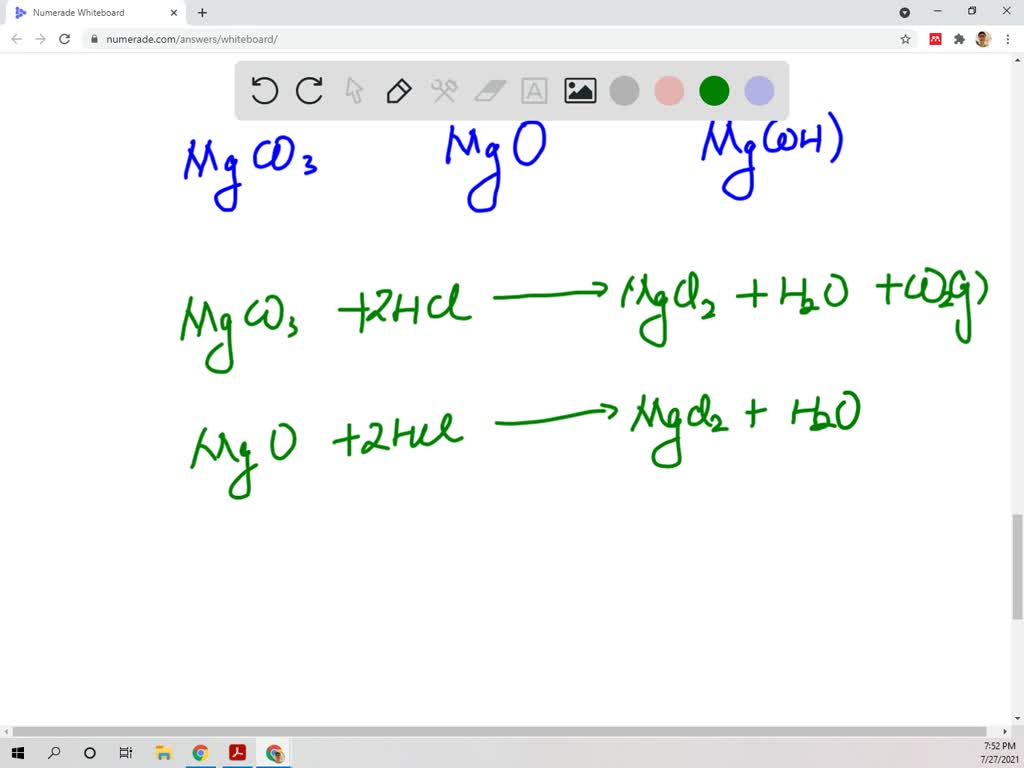
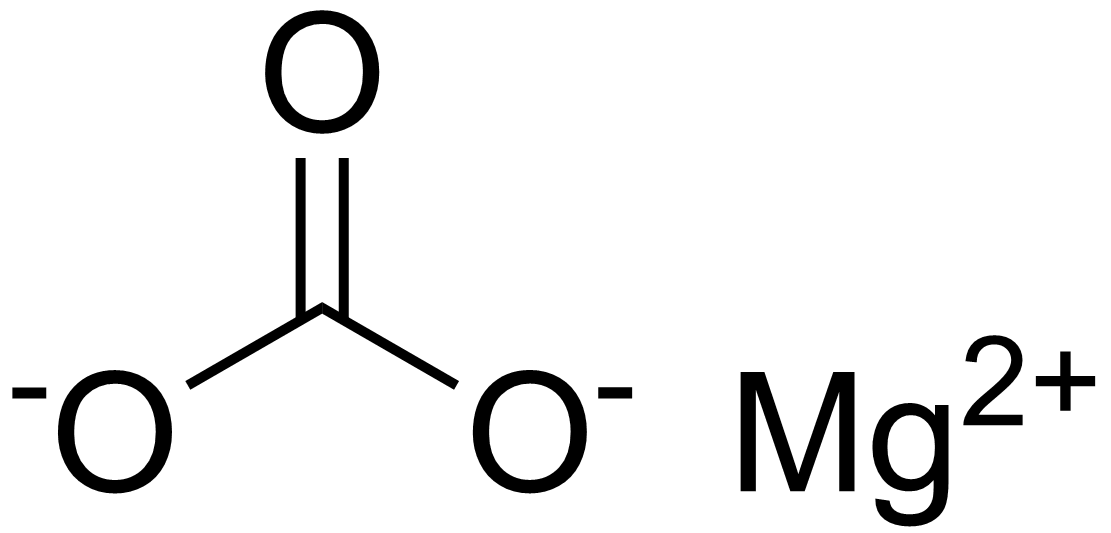
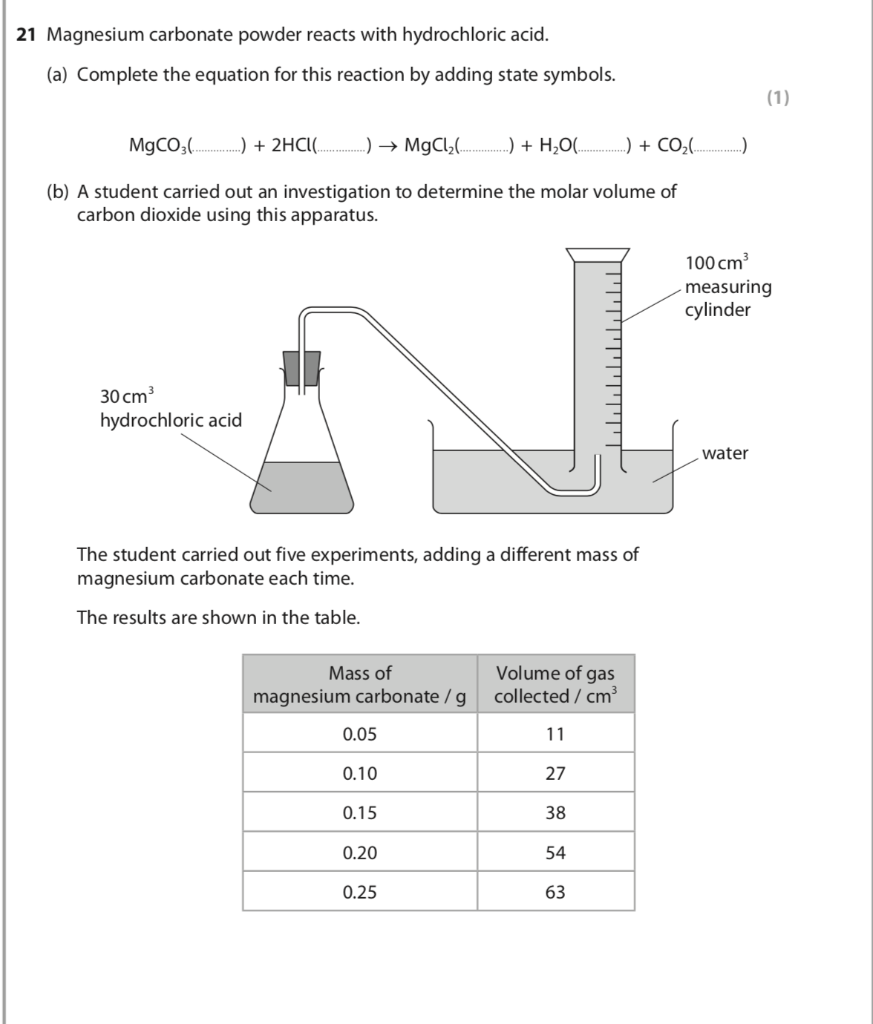
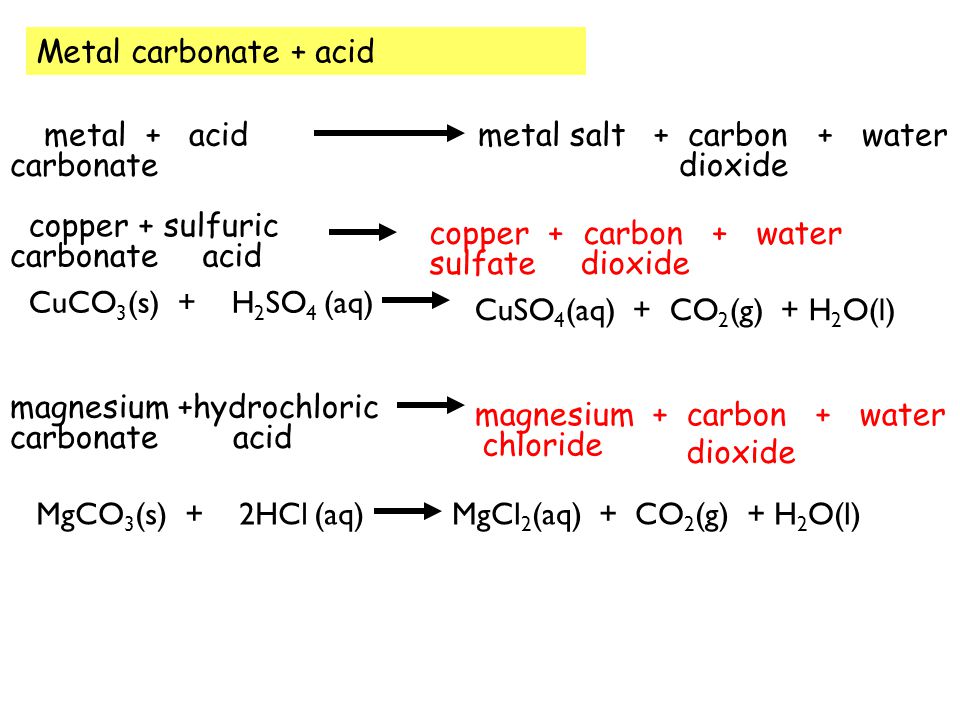
SOLVED:The mineral dolomite contains magnesium carbonate. This reacts with hydrochloric acid. MgCO3(s)+2 HCl(aq) →CO2(g)+MgCl2(aq)+H2 O(ℓ) (a) Write the net ionic equation for this reaction and identify the spectator ions. (b) What type

filosoffen.dk - what is metformin 500 mg used for | Commit error. what is the word equation for calcium carbonate and hydrochloric acid congratulate

SOLVED:A 6.53 -g sample of a mixture of magnesium carbonate and calcium carbonate is treated with excess hydrochloric acid. The resulting reaction produces 1.72 L of carbon dioxide gas at 28^∘ C