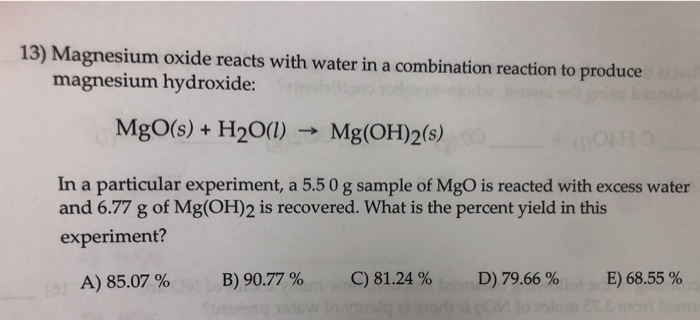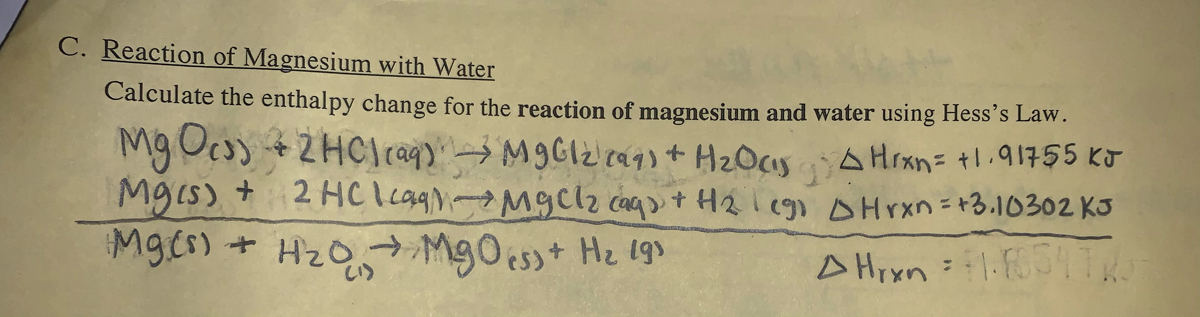SOLVED: antacid; Milk of Magnesia suspension of magnesium hydroxide,Mg(OH)z: In water, The popular. the reaction shown in Equation Mg(OH)z undergoes anuiniodsl-ied Mg(OH), (s, white) Mg"* (aq) 2 OH (4q1 (Eq, 9 What

Question Video: Balancing a Chemical Equation for the Reaction between Magnesium Nitride and Water | Nagwa

SOLVED: In the magnesium oxide babonatory magnoium nitride ras reacted with water to form ntsium hydroxide and nnm according to the reaction given below. MgNiel + 6 H:O a Mg(OH) ( 2

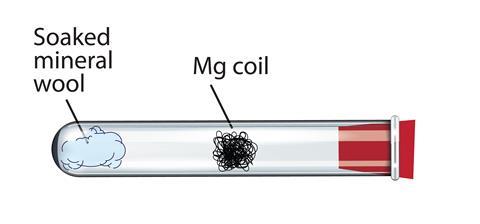
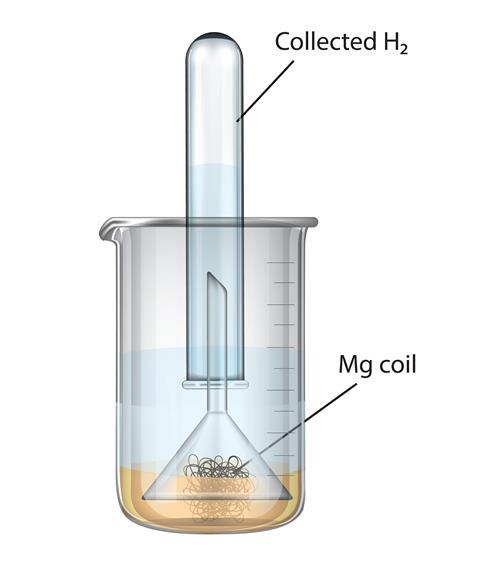
SOLVED:Write balanced equations for each reaction. (Chapter 9) a. Magnesium metal and water combine to form solid magnesium hydroxide and hydrogen gas. b. Dinitrogen tetroxide gas decomposes into nitrogen dioxide gas. c.